 Potassium Cold Water Potassium Hydroxide Hydrogen The reaction between metals and water – Metals from potassium to calcium can react with cold water and release hydrogen gas.Ĭhemical Equations for the reaction of K and Ca with cold water are: So, by reactivity series, you can tell which metal will displace another metal. In displacement reaction – Displacement reactions are those reactions in which more reactive metal displaces less reactive metal from its salt. When we move down the series the electron-donating capacity of metals decreases. While metals from Zinc to Hg can be extracted by simply reducing their oxides, which is an inexpensive method. The metals which are placed above in the series can be extracted by electrolysis. Thus, metals placed at the top of the reactivity series can remove the metals which are present at the bottom of the series from their salts. The metal which is more reactive than other metals can remove less reactive metal from its salt. Metals present in the reactivity series above hydrogen can remove hydrogen ions from dilute HCl or Dilute sulphuric acid. Thus, potassium is the strongest reducing agent.Īs we go down the reactivity series, the ability of metals to remove hydrogen from hydrides decreases. The reducing power of metals decreases as we go down the series. The electropositive character of metals decreases as we go down the series. Metals present at the top of the reactivity series are highly electropositive metals. May react with some of the strong oxidizing acids May react with some strong oxidizing acids Reacts with acids very poor reaction with steam Or less commonly other alkali metals, hydrogen or calcium in the Kroll process 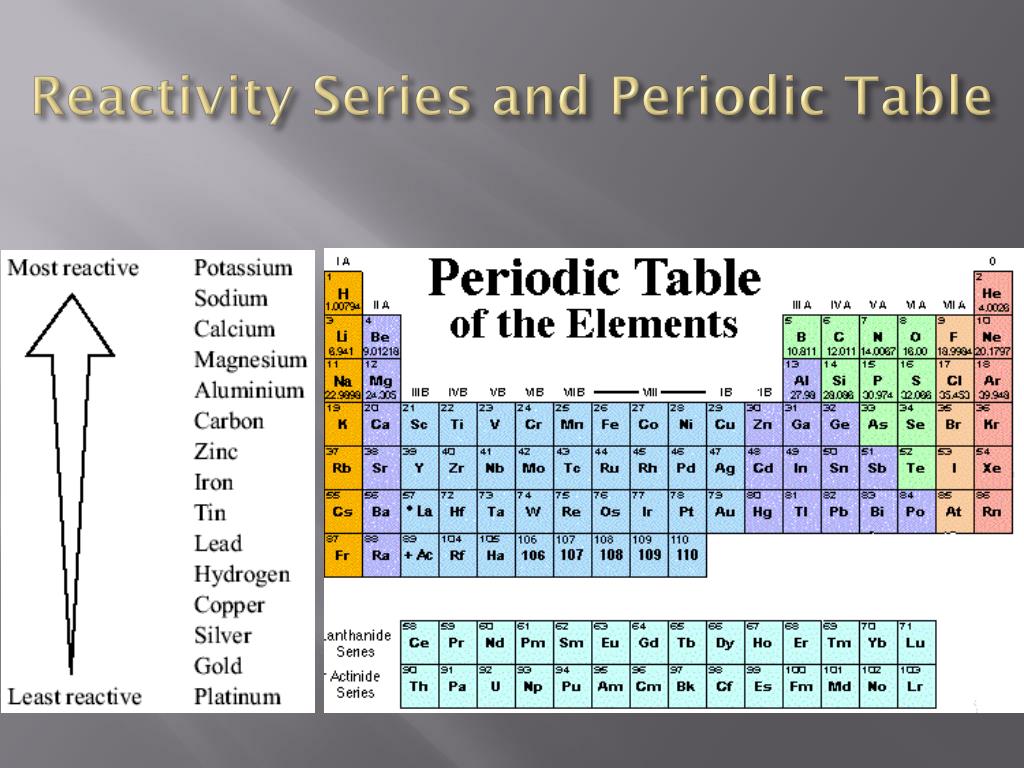
Pyrometallurgical extraction using magnesium, In boiling water, and very vigorously with acids. Reacts very slowly with cold water, but rapidly Here is the tabular format of the same.Įlectrolysis (a.k.a. Here metals are segregated in terms of their reaction with cold water, hot water, acid, and steam and reaction with concentrated mineral acids. The reactivity of the metals is given below in another tabular format where it has been mentioned along with its ions. Hydrogen is a non-metal but still, it has been included in the reactivity series as it helps in the comparison of reactivity of metals. While metals such as zinc, aluminium, magnesium, calcium, etc. This is the reason why platinum and gold don’t get corrode easily and don’t form oxides. Metals from copper to platinum are highly unreactive and don’t react with any other substance in normal conditions. While metals from magnesium to lead can react with acids. It is important to recognize which numbering system is being used and to be able to find the number of valence electrons in the main block elements regardless of which numbering system is being used.Metals from potassium to calcium are highly reactive and even react with water. You will come across periodic tables with both numbering systems. In this numbering system, group 1A is group 1 group 2A is group 2 the halogens (7A) are group 17 and the noble gases (8A) are group 18. The elements in this group are also gases at room temperature.Īn alternate numbering system numbers all of the \(s\), \(p\), and \(d\) block elements from 1-18. We will learn the reason for this later, when we discuss how compounds form. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. This group contains very reactive nonmetal elements. Group 7A (or 17) elements are also called halogens. It is important to recognize a couple of other important groups on the periodic table by their group name. Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table. The same pattern is true of other groups on the periodic table. 
Once again, because of their similarities in electron configurations, these elements have similar properties to each other. Group 2A is also called the alkaline earth metals. Although most metals tend to be very hard, these metals are actually soft and can be easily cut. 
Group 1A is also known as the alkali metals. Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
